|
|
|||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
Powered by: Sun Microsystems |
|
Battery
Products
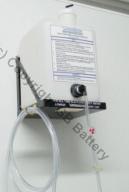
Example: Forklift Battery Watering System
Automatic Watering System
| Filler is Easily
Accessible | 5gal.
Reservoir | Water Level Indicator
The five most common
reasons for Premature Battery Failure: Low water levels
· From our experience, this is the number one cause of battery failure. ·
Charging batteries that are as
little as 1” low on water can cause damage to the entire battery that can
never be repaired. It dries and burns the uppermost portion
of the “plates”, causing high resistance that is permanent, and
effectively isolates the portion that remained submerged. ·
Even when the proper water levels are restored, the damaged
portion continues to cause high resistance and the battery runs much hotter
from that point forward, causing accelerated water loss and further plate damage due to overheating. Over Charging and
Opportunity Charging
·
Industrial batteries are typically
designed to last at least 1,500 charge cycles, over
a five to fifteen year period. Each time you charge a battery, regardless of
how long, it constitutes one cycle. ·
Consistently charging a battery
twice per day, during lunch breaks for example, is known as Opportunity
Charging, and reduces the useful life of a battery by 50%. ·
The additional heat generated by opportunity
charging a battery usually reduces the run time equal or greater in
proportion to the amount of charge it actually received, making the practice
completely ineffective and costly. ·
Routinely charging the battery
before it is 80% discharged is another common form of over
charging. For example, if you only use the battery a few hours a day, it’s
best to use it until it is truly in need of charging before actually plugging
it in. Remember, each charge constitutes one cycle, so try not to charge
unnecessarily. Over watering
·
Commonly occurs as a reaction to low
water levels, but is a major problem unto
itself. ·
Flushes the electrolyte from the cells and gradually dilutes the acid to the point that the battery can no
longer function properly. In many cases this can be remedied sending the
battery out to have the acid adjusted, but the battery’s life will still be
shortened somewhat. ·
Causes tray corrosion (listed
below). Failing to Equalize Charge
·
Batteries should receive an Equalize
Charge once every 10 cycles. ·
Almost all chargers are equipped
with an Equalize feature. On older chargers, this setting is usually referred
to as Weekend or Weekly charge. ·
Selecting this setting adds 3
additional hours to a normal charge, ensuring all of the cells in the battery reach full charge, and
the allowing extra time for the electrolyte to mix during the Gassing
Stage. ·
Failure to equalize causes reduced
battery run time and eventual failure, due to Sulfation, Stratification, and
an imbalanced capacity between the cells. Battery Corrosion
·
Batteries should be rinsed or washed
at least once per year to prevent corrosion. ·
Even when the proper water levels are consistently
maintained, sulfuric acid vapors escape during charge.
These vapors leave an oily sulfuric acid residue on the top of the
battery around the vent caps. Over time, the water in the residue evaporates
leaving full strength, concentrated acid that is much more concentrated than
the diluted acid inside the battery. ·
The concentrated acid is very
conductive. As it gradually accumulates and spreads out, it eventually makes contact
between the intercell connectors across the top of the battery. This results
in shorting between the cells, causing the battery to self-discharge, and additional heat during charge
and use. ·
Even though battery trays have a
baked on powder coat finish, they will easily corrode if the residue is not
rinsed off. The corrosion will become progressively worse until it is either
removed, or it destroys the battery. |
|||||||||||||||||||||||||||||||||||||||||||
|
|
|||||||||||||||||||||||||||||||||||||||||||||||
CABLE CORROSION
– REPAIRABLE:
This is a commonly overlooked problem which causes reduced
run time and all of the classic symptoms of a bad battery or cell. Many times this is the only problem,
and replacing the corroded cable increases run time dramatically. ·
CAUSE:
If the battery is not rinsed periodically, acid penetrates the seal between the cable
head and the insulation and begins to oxidize and corrode the copper
conductors. The cable gradually swells over time as the copper breaks down,
deteriorating the connection gradually until the battery no longer accepts a
charge. Examples of Battery Cable Corrosion: Swollen near
terminal, copper is replaced by blue corrosion.
Positive Plate Growth
– NOT REPAIRABLE:
The true sign that a battery has reached the end of its
useful life, and is in need of replacement. Typically begins to occur after 5
years. As batteries age, the positive plates begin to soften and expand in size.
This causes internal pressure inside the battery cells. Most manufacturers now use floating
bushings on the positive cell posts to allow for this gradual
expansion and reduce stress on the cells internal components. Eventually, as
the battery reaches the end of it’s normal useful
life, the internal pressure is great enough to push the positive posts well
beyond normal tolerances and can begin to distort the cell covers and vent wells. Positive plate growth is normal and should be
considered as a wear indicator. If your battery is no longer holding a charge
and shows signs of plate growth, it should be replaced, not
repaired. ·
Batteries can fail and be beyond
economical repair without showing noticeable signs of plate growth. However, plate growth is a sure, visible sign that
the battery is beyond economical repair. ·
Positive plate growth typically occurs after 5 years
or 1,500 cycles. If it occurs in less than 5 years, it is most likely due to
opportunity charging or over charging, which should be addressed when
replacing the battery. ·
This condition usually affects the Positive posts only, and
will give an uneven, slanted appearance when viewed from the side, looking
across the top of the battery. Examples of Positive Plate Growth:
·
SHORT
RUN TIME: Progressively shorter run times could be
caused by a malfunctioning battery OR charger. It is best to have both
checked if you begin to experience shortened run time or suspect a problem. ·
EQUALIZE
CHARGE: Industrial batteries must receive an
equalize charge at least once per month, or permanent damage will result.
Never equalize more than once every 5th charge cycle, or damage will result. Reason:
Selecting equalize mode on your charger adds 3 additional hours of charge
time to that charge cycle, ensuring all of the cells reach 100% charge. Equalizing too
often or too little will shorten battery life. ·
WHEN TO
ADD WATER, AND HOW MUCH TO ADD: Water can be
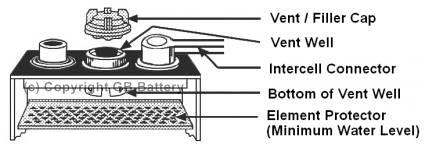
added before or after charging. Add just enough water to cover the perforated
element protector (visible at the bottom of the vent well). ·
·
WHAT
KIND OF WATER TO USE: In general, normal city water will
suffice, but if you have any doubts about purity of the local water supply,
A) contact the GB battery supplier in your area B) get a chemical analysis of
the water C) use distilled water. ·
·
SPECIFIC GRAVITY (SPGR): See SPGR chart
below. ·
OVER DISCHARGING: Never over
discharge batteries. More is not necessarily
better when it comes to recharging batteries. Most battery manufacturers
warrant their batteries for up to 1,500 cycles of charge and discharge provided, among other things, that
the battery is never discharged beyond 80%. This normally
coincides with an eight-hour shift. Trucks fitted with extra equipment such
as clamps, high speed lifts, etc. will need a higher capacity battery to ensure the battery is
not discharged beyond 80%. Lift truck
interrupts are available to detect the correct discharge level and are recommended by battery
manufacturers as a means of ensuring batteries are not over discharged. The best way to ensure batteries
are not being overcharged is to periodically (once a
month) check the temperature of the center cell on a battery at the end of regular
charge. If the temperature of the electrolyte is more than
36° F above the ambient temperature, call your battery technician—
there is a problem. ·
AVOIDING SPARKS: Batteries produce
and store hydrogen gas, which is highly explosive. Never weld near a battery.
Never place metal objects on batteries. Such
objects can cause a short circuit between adjacent cells and result in possible injury to those
close to the battery. Similarly, people charged with caring for or operating
batteries should not wear any metal jewelry. ·
UNDER-SIZED CHARGERS: A charger that is
more the 100AH less than the your battery’s rated capacity.
Will result in an undercharged battery with significant reduction in
operating life. Your local GB vendor has all the information to ensure the
battery and chargers are precisely matched. ·
OVER-SIZED CHARGERS: A charger that is
over 100AH more than your battery’s rated capacity.
Can overcharge and overheat your battery, reducing the operating
life of your battery. ·
WHEN TO
REPLACE A BATTERY: Repair or replace batteries when capacity that has fallen below 80% of its
rated capacity. Continuing to operate a bad
battery can damage a truck's electric motor and electronics. Failing
batteries also require recharging more frequently, wasting hundreds or thousands
of dollars in energy per year, depending on the size of your fleet. ·
RECORD
KEEPING: We recommend that each forklift, battery
and charger in your fleet be given a unique number for easy identification
and tracking purposes. Keep regular records on the maintenance of batteries.
For instance, keep a log of ever time a battery is watered and equalize
charged, or at minimum, each time an operator complains of short run time and
whenever cells or cables are replaced. These records
will be invaluable when it comes to predicting when battery replacement will
be necessary. ·
FORMULAS:
§ Charger Model# Breakdown: Example, Model GB100-3-18-750 = GB brand, 100% rated, 3 phase, 18 cell (36 volt), 750AH (Ampere Hour). § Battery
Model# Breakdown: Example, Model 18-125-13 = 18 cells (x2 = 36 volt), 125AH per plate, 13 negative plates. § Calculating
AH Capacity: Negative plates per cell – 1, / 2 x AH per plate = AH (Ampere Hour). § Example,
18-125-13: 13 – 1 = 12, / 2 = 6 x 125 = 750AH § Calculating
KWH Capacity: AH x
Volts (2v per cell) = WH (Watt Hour) / 1000 x .97
(efficiency factor) = KWH (Kilo Watt Hour) § Example,
battery model 18-125-13, 36V / 750AH: 750 x 36 = 27,000 / 1000 = 27 x .97 =
26.19KWH. ·
STATE OF CHARGE: Typical
no-load voltages vs. state of charge:
§ Sample
voltages are for a 12-volt / 6 cell battery system (figured at 10.5 volts =
fully discharged, and 77 degrees F). For a 24-volt
battery multiply by 2, for a 36-volt battery multiply by 3, 48-volt battery
multiply by 4. VPC is the volts per individual cell - if you measure more than a .2 volt
difference between each cell, you need to equalize, or the battery
is beginning to go bad, or it could be sulfated. These voltages are for
batteries that have been at rest for 3 hours or more. Batteries that are
being charged will be higher - the voltages while charging will not tell you
anything, you have to let the battery sit for approx. 3hrs. For longest life,
batteries should stay in the green zone. Occasional dips into the
yellow are not harmful, but continual discharges (or “cycles”) to those levels will shorten
battery life considerably. It is important to realize that voltage
measurements are only approximate. Note the large voltage drop in the
last 10%.
Other Battery Questions and Answers
How are batteries rated and what
do the ratings mean in battery selection? How does the Cold Cranking Amperage rating help me select
a battery? What does the Reserve Capacity rating mean and how does
it apply to deep cycle batteries? What is
the difference between deep cycle batteries and starting batteries? Does overcharging damage batteries? Does over discharging damage batteries? How can I evaluate the health and state of charge (SOC)
of a battery?
1) BATTERY
RATINGS
|
|
||||||||||||||||||||||||||||||||||||||||||||||
Fork truck battery for sale, forklift batteries for
sale
Forklift Batteries New York NY
Forklift batteries New Jersey NJ
Reconditioned forklift batteries, Lift Truck batteries
Industrial forklift batteries, Industrial forklift
battery chargers
Forklift battery Orlando Florida FL
Forklift battery Los Angeles California CA
Forklift Battery, Lift Truck Battery, Fork Lift
Battery, Fork Truck Battery
Forklift battery, Lift Truck Battery Chicago Illinois
IL
Lift Truck Battery Pennsylvania PA
Phoenix Forklift Battery Arizona
Fork Truck Batteries, Forklift Batteries
Forklift Batteries Inc, Batteries Inc Maryland MD
Lift Truck Batteries, Fork Lift Batteries
Forklift battery Atlanta Georgia GA
West Michigan Battery, Forklift Batteries
Extra Power Battery, Xtra Power Battery
Texas Industrial Battery, Lift Truck Battery TX
Forklift Battery, Dallas TX, Houston TX, Austin TX
Forklift Battery NJ New Jersey
http://forkliftpower.com/6-85-11.com/index.html
http://forkliftpower.com/6-85-13.com/index.html
http://forkliftpower.com/6-100-13.com/index.html
http://forkliftpower.com/6-125-13.com/index.html
http://forkliftpower.com/24v-battery.com/
http://forkliftpower.com/12-85-5.com/index.html
http://forkliftpower.com/12-85-7.com/index.html
http://forkliftpower.com/12-85-13.com/index.html
http://forkliftpower.com/12-85-7.com/index.html
http://forkliftpower.com/12-85-19.com/index.html
http://forkliftpower.com/12-85-21.com/index.html
http://forkliftpower.com/12-85-23.com/index.html
http://forkliftpower.com/12-100-13.com/index.html
http://forkliftpower.com/12-125-11.com/index.html
http://forkliftpower.com/12-125-13.com/index.html
http://forkliftpower.com/12-125-15.com/index.html
http://forkliftpower.com/12-125-17.com/index.html
http://forkliftpower.com/36v-battery.com/
http://forkliftpower.com/18-85-13.com/index.html
http://forkliftpower.com/18-85-15.com/index.html
http://forkliftpower.com/18-85-17.com/index.html
http://forkliftpower.com/18-85-19.com/index.html
http://forkliftpower.com/18-85-21.com/index.html
http://forkliftpower.com/18-85-23.com/index.html
http://forkliftpower.com/18-85-25.com/index.html
http://forkliftpower.com/18-85-27.com/index.html
http://forkliftpower.com/18-85-29.com/index.html
http://forkliftpower.com/18-85-31.com/index.html
http://forkliftpower.com/18-85-33.com/index.html
http://forkliftpower.com/18-125-11.com/index.html
http://forkliftpower.com/18-125-13.com/index.html
http://forkliftpower.com/18-125-15.com/index.html
http://forkliftpower.com/18-125-17.com/index.html
http://forkliftpower.com/48vbattery.com/index.html
http://forkliftpower.com/24-85-13.com/index.html
http://forkliftpower.com/24-85-15.com/index.html
http://forkliftpower.com/24-85-17.com/index.html
http://forkliftpower.com/24-85-19.com/index.html
http://forkliftpower.com/24-85-21.com/index.html
http://forkliftpower.com/24-85-25.com/index.html
http://forkliftpower.com/24-85-27.com/index.html
http://forkliftpower.com/24-125-13.com/index.html